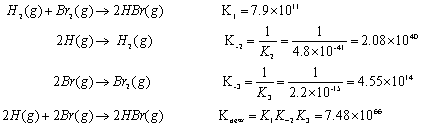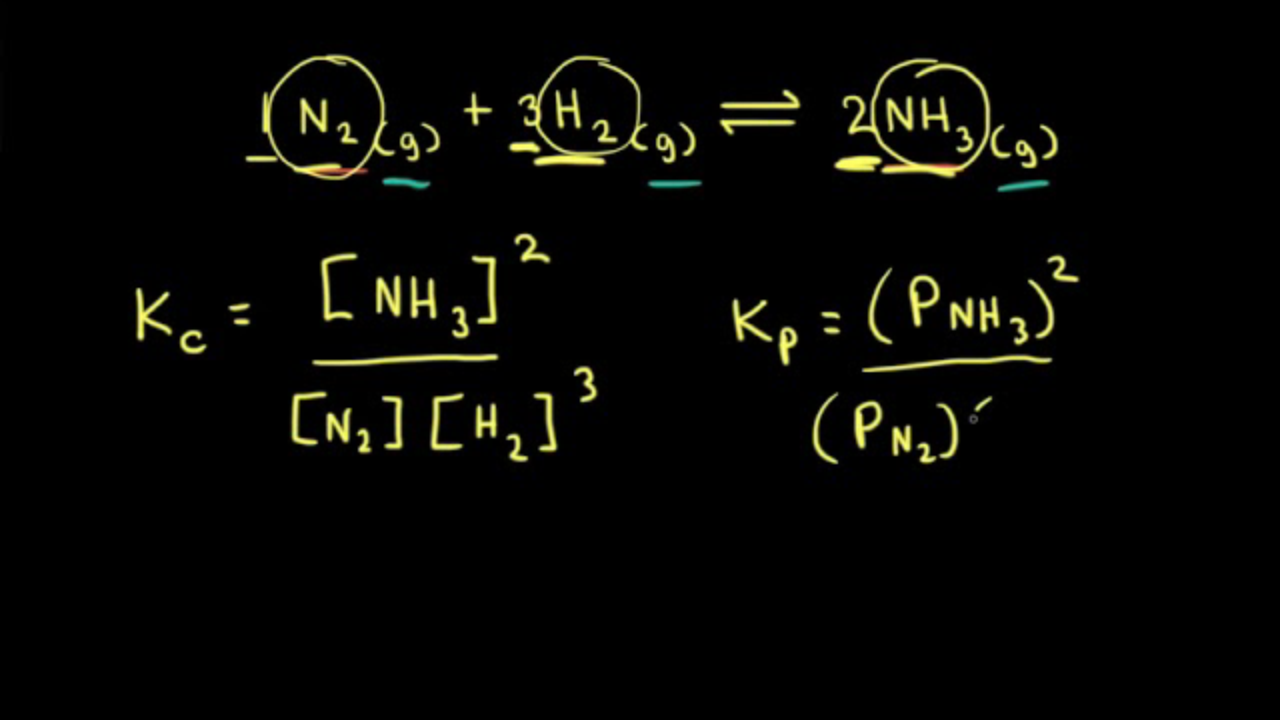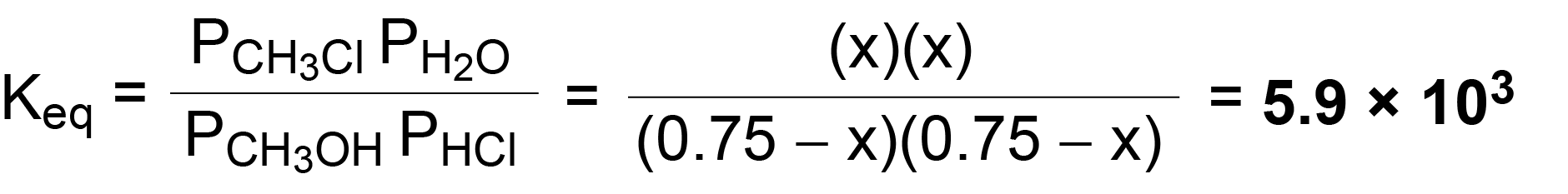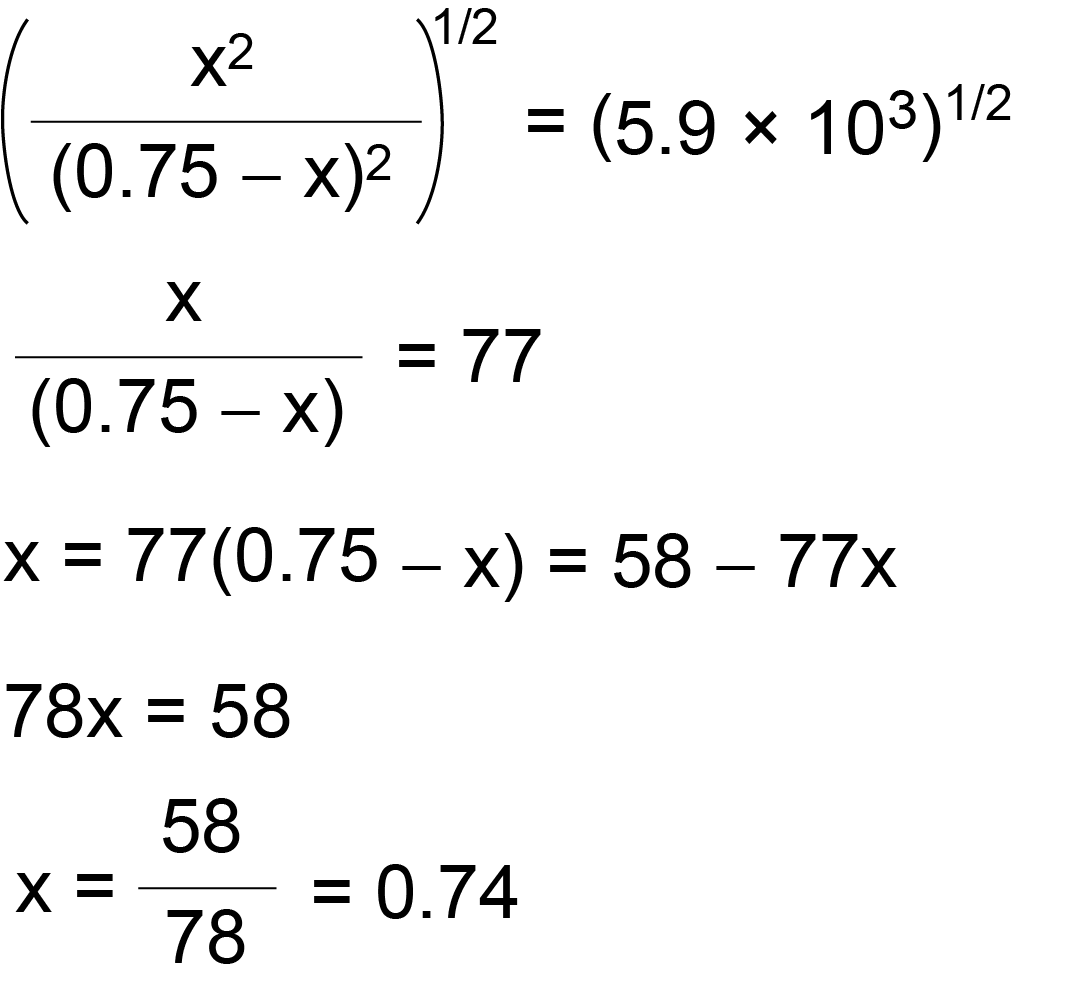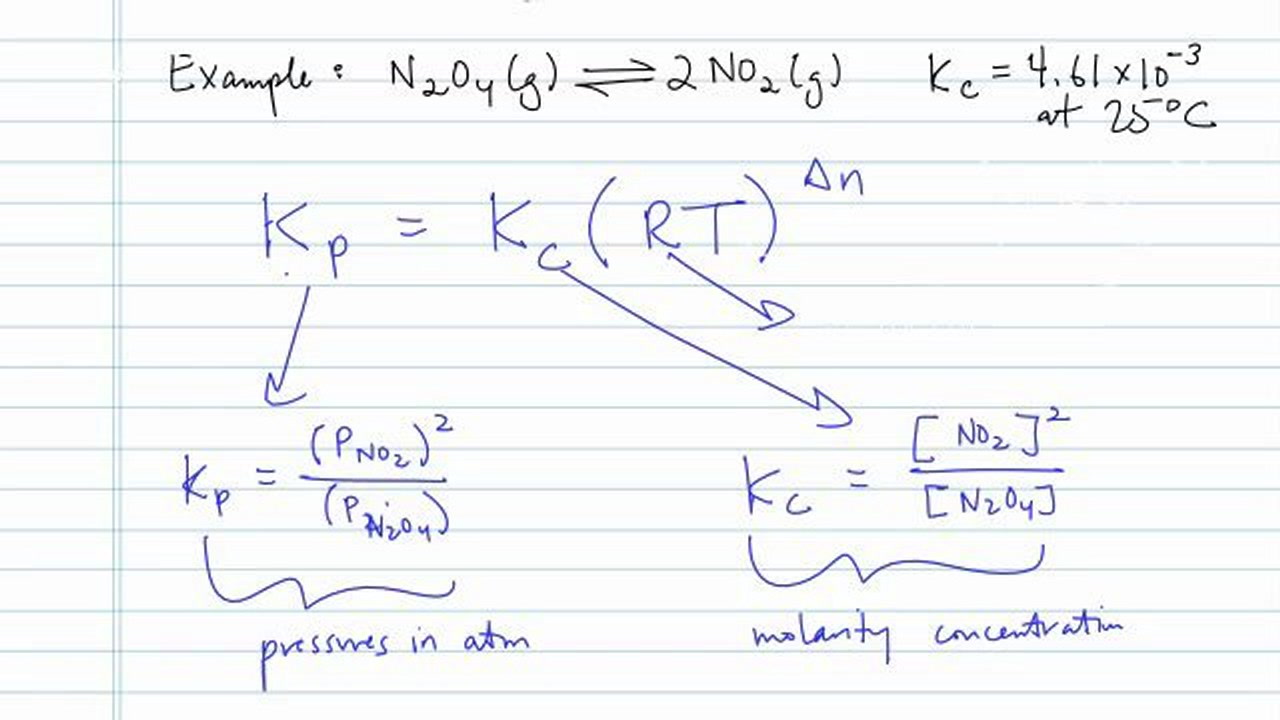SOLVED:The reaction CO2(g) + C(s) 2 CO(g) has Kp = 5.78 at 1200 K. a. Calculate the total pressure at equilibrium when 4.45 g of CO2 is introduced into a 10.0-L container

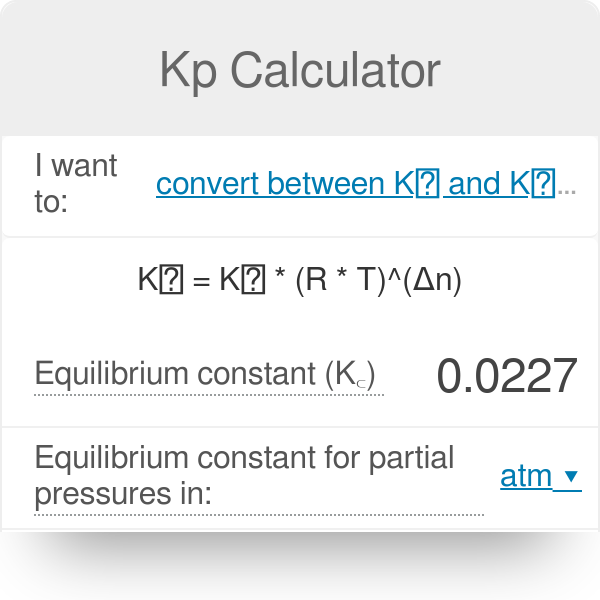
SOLVED: Tutored Practice Problem 15.2.2 counistownrdsGrndp Interconvert Kp and Kc values: Calculate Kp for the following reactions at the indicated temperature. PCls(g) PClz(g) + Cl(g) Kc 1.42*10-2 at 504 K Kp (b)

Learn how the equilibrium constants Kc and Kp are related. | Chemistry help, Ap chemistry, High school chemistry
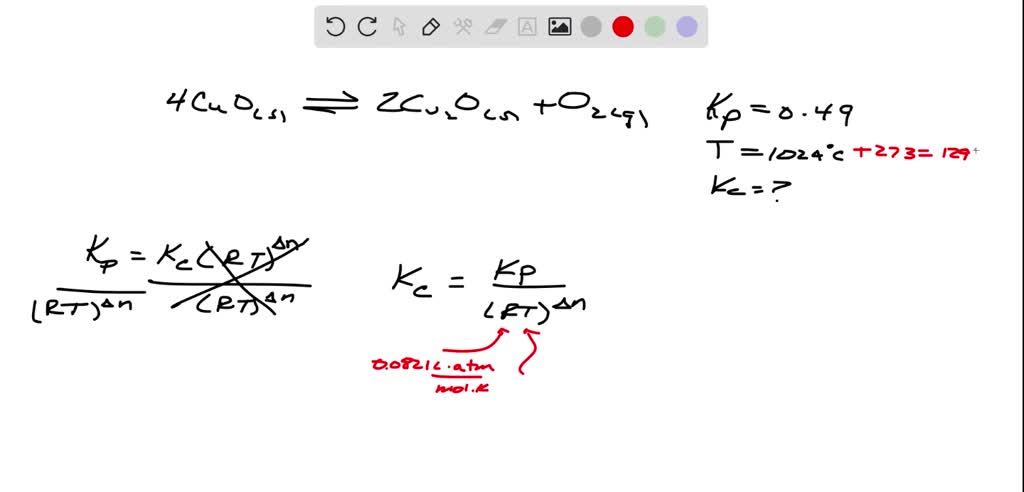
SOLVED: Kp for the reaction: 4CuO (s) <—-> 2Cu2O (s) + O2 (g), is 0.49 at 1024 ° C. Calculate Kc a this temperature.
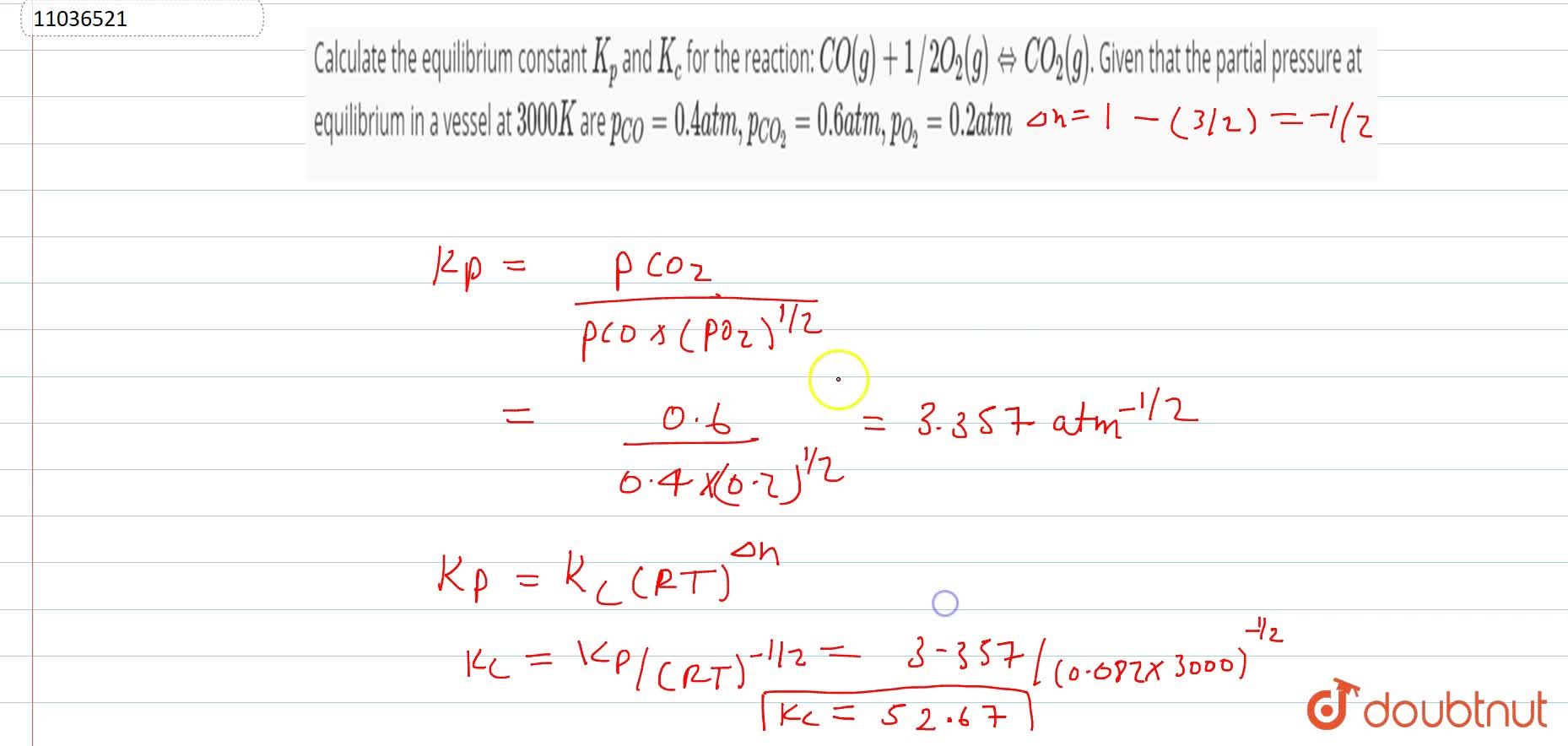
Calculate the equilibrium constant K(p) and K(c ) for the reaction: CO(g)+1//2O(2)(g) hArr CO(2)(g). Given that the partial pressure at equilibrium in a vessel at 3000 K are p(CO)=0.4 atm, p(CO(2))=0.2 atm